Thousands of people have total hip replacement surgery every year, and while the vast majority of them return to normal life without complications, there are times when complications arise. Some complications are a result of the surgery itself, while others are related to the artificial joint used to replace the damaged hip. The U.S Food and Drug Association (FDA) recall of one such device has raised concerns over the safety of its manufacturing process, as leftover residue on the implants was the reason for the recall. Although the Zimmer Kinectiv prosthesis is designed to help patients, if the device causes injury affected patients may be eligible to pursue claims against the device maker with the assistance of an Iowa Zimmer Kinectiv lawsuit attorney.
If you or a loved one is experiencing problems with a Zimmer hip implant or any metal-on-metal hip replacement, contact Attorney Group for Iowa today. We provide free, confidential, no-obligation consultations, and if you have case we can connect you with an affiliated Zimmer Kinectiv lawsuit attorney who can assist you throughout the legal process. The time to pursue a claim is limited, so contact us today.
When Do Doctors Choose Hip Replacement Surgery?
For those dealing with hip pain, practically every day-to-day activity can be unbearable. Many physicians attempt to treat hip pain without a surgical procedure before turning to a hip replacement. The most common types of non-surgical treatments include the use of cortisone shots, anti-inflammatory drugs, weight loss, walking aids or physical therapy. If these types of treatment are ineffective, then doctors typically suggest surgery.
According to the National Institute on Aging, the most common reason for a patient to have a total hip replacement is osteoarthritis. This condition causes the cartilage within the hip joint to break down, which means that the bones are rubbing together. This can lead to severe pain, as well as irreversible damage to the joint. Today, surgeons typically decide whether or not a person is a candidate for surgery based upon general health and pain levels, rather than age.
Hip Replacement Challenges
One challenge that many surgeons face when performing hip replacement surgery is how to restore hip offset and leg length. Some implants limit the amount of adjustability during surgery, which can lead to a limp, pain or dislocation later on. Implants such as the Zimmer Kinectiv M/L Taper Hip Prosthesis are designed to allow surgeons to adjust leg length and hip offset in order to avoid future issues. Zimmer claims that its product gives surgeons control over leg length adjustment based on an individual patient’s gender and anatomical make up.
A study performed by researchers at The New Mexico Center for Joint Replacement Surgery found that hip replacement implants with two modular junctions, like the Zimmer Kinectiv device, resulted in the greater ability of surgeons to restore leg length than with implants with only a singular junction. However, this benefit could be outweighed by other concerns with the devices, such as the potential for mechanical failure, increased amounts of corrosive debris and increased cost.
Zimmer Kinectiv M/L Taper Recall
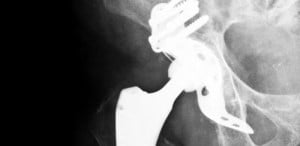
In June 2015, the FDA issued a recall of the Zimmer M/L Taper with Kinectiv Technology Femoral Stems and Necks. In May of the same year, the manufacturer notified the FDA of an issue with process monitoring that led to manufacturing residue being left on the devices. These prostheses are made of titanium alloy, and the FDA states that the residue could cause serious medical complications in those patients who received the tainted implants.
The FDA lists pain, infections, allergic reactions and death as possible health concerns connected with the residue. Any patients who underwent surgery and received one of the devices that were manufactured and distributed between March 31 and April 20, 2015, may require revision surgery to change the implant out. The FDA has asked medical professionals and patients to report any problems to the agency through their MedWatch Safety Information and Adverse Event Reporting program.
Affected Patients May Be Eligible to File An Iowa Zimmer Kinectiv Lawsuit
Device makers have a duty to design and produce safe products, and to warn of possible risks associated with their products. Failure to fulfill that duty can result in injuries to patients, and the device maker being held liable for those injuries.
Patients who are injured by metal-on-metal hip replacements may be entitled to compensation for damages resulting from injuries. Compensation can be based on factors including:
- Past and future medical expenses
- Pain, suffering, and mental anguish from an injury
- Loss of income or ability to work due to loss of mobility
If a loved one dies after complications from a metal-on-metal hip implant, family members may be able to pursue claims for wrongful death damages, including:
- Conscious pain and suffering of a loved one prior to death
- Loss of financial support
- Pain, suffering, and mental anguish resulting from the loss of a loved one
Affected patients and their families are encouraged to seek the advice of an Iowa Zimmer Kinectiv lawsuit attorney to discuss their legal rights and options for compensation.
Contact Us For More Information
If you have been injured by a metal-on-metal hip replacement, or to find out more about an Iowa Zimmer Kinectiv lawsuit, contact Attorney Group for Iowa. You can fill out the form on this page, call us at the number listed at the top of the page, or email us at info@attorneygroup.com.
When you contact us, an attorney will follow up with you to speak with you about your case or answer questions that you might have. There is no cost or obligation to speak with us, and any information you provide will be kept confidential.
Please note that the law limits the time you have to pursue a claim for an injury. If you think you have a case, you should not delay taking action.
See our Frequently Asked Questions page for more information, and contact Attorney Group for Iowa today.





