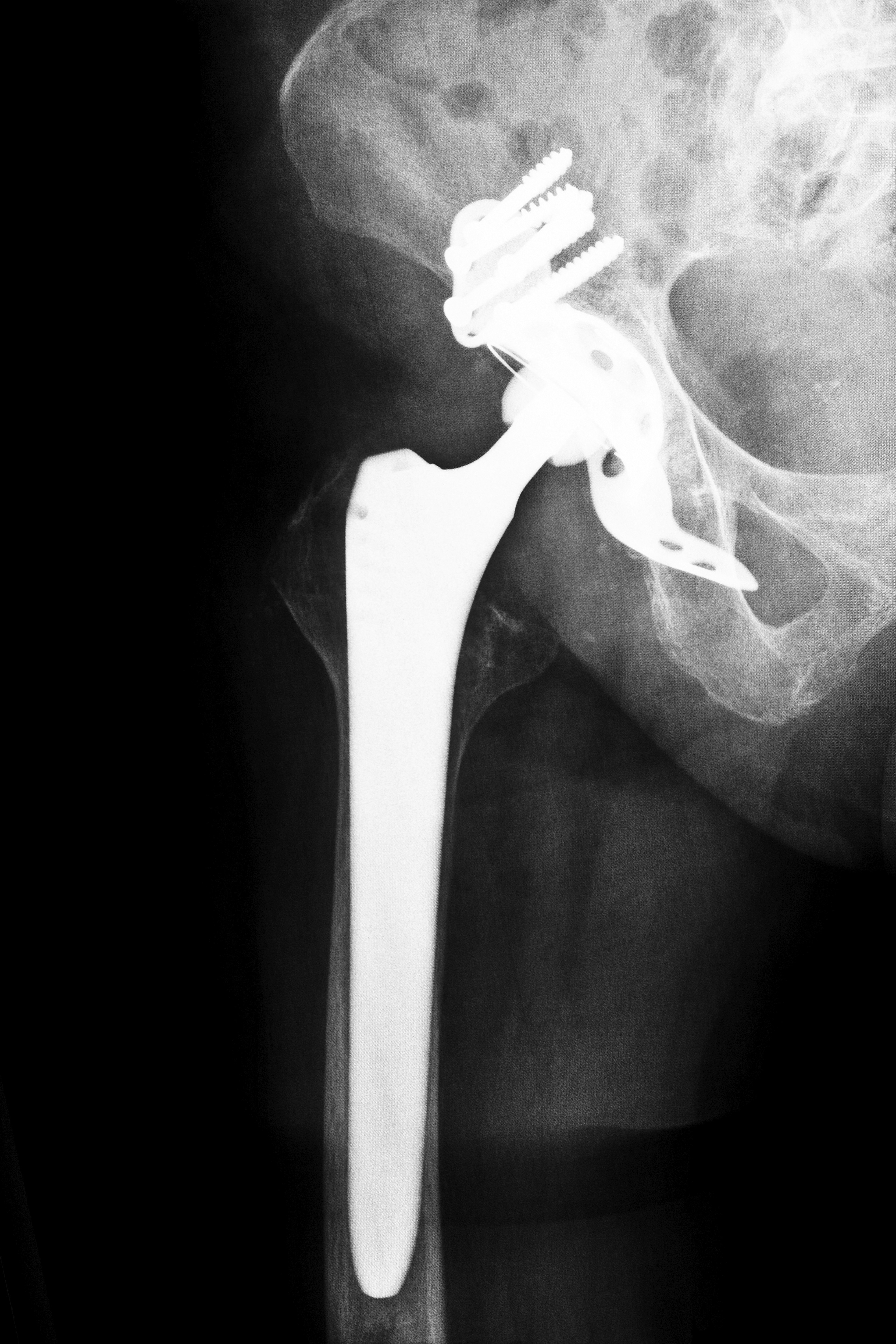
The FDA recently announced that Stryker Orthopedics had issued a recall for two of their implants voluntarily. The implants are: the “ABG II Modular Neck Hip Stem” and “Rejuvenate Modular” which were used in hip transplant surgery. In July 2012, the corporation announced that it would remove these two products from the market. The company stated that corrosion can happen in these two devices, and that metal debris can be released by the wear and tear process which can then lead to swelling, pain and other adverse tissue reactions. While the hip sockets of these two models are not comprised of metal on metal joints, the recalled implants do have a metal on metal joint built within them. This means that the implants can shed small particles of metal as they deteriorate over time. The recipients of these implants can develop a condition called Metallosis, which usually results from the harmful accrual of metal fragments into the body’s soft tissues. In order to reduce and alleviate the swelling and pain brought about by metallosis, patients with the Stryker hip implants may have to undergo painful corrective surgeries and hospitalization.
In their announcement, Stryker Orthopaedics encouraged the recipients of these two implants to contact their surgeons. Stryker Orthopaedics makes various medical implants and devices, but the recent recall specifically targets these two:
- The ABG II Modular Neck Hip Stem
- The Rejuvenate Modular Hip System
If you are unsure of the specific implant you received, it is advisable to check your medical records or contact your surgeon.
What The Stryker Orthopaedics Means For You and Your Loved Ones
The Stryker Rejuvenate Modular Hip system uses a specific design of a ceramic ball and socket and not the metal on metal implant which has in the past caused a lot of media media scrutiny and attention. However, this particular implant has a joint that contains metal parts which can be dangerous as the device deteriorates over time. Some recipients of this implant have been reporting major swelling and pain at their hip joints.
Besides causing the patients a lot of discomfort, the major concern is the long term undesirable effects these implants will have on the recipients. The ongoing corrosion and the wear and tear on the metal component can cause debris to chip off and enter the bloodstream, ultimately getting into the liver, kidneys, spleen and lymph nodes. As these particles accumulate in the tissues and blood, metal toxicity can occur.
Metal toxicity (metallosis) is a grave medical condition that can cause damage to the soft tissues and the bones. This condition can also lead to loosening of the hip implant and cause its ultimate failure. Some recipients of these implants have been reporting of their failure to work after a relatively short period (5 years after the surgery) whereas they were supposed to function for at least 15-20 years. When hip implants fail, medical surgery is often required to correct the problem.
Other side effects that may occur because of continued exposure to metals may include gastrointestinal problems, headaches, dizziness, emotional problems, confusion, and a tingling, numb or burning sensation in the arms, hands, feet and legs. There is also some anxiety about long term damage to the patient’s chromosomes and DNA.
What You Should Do
If the recalled Stryker hip implant was used in your surgery, if you underwent hip replacement surgery recently, or if you are experiencing swelling and pain around your hip region, it is advisable that you make an appointment with your surgeon as soon as possible. Your surgeon will be able to tell you the specific type of implant you received. You can also ask for copies of your medical records and find out. Stryker Orthopedics have established a call center where you can call and make further inquiries at 1-888-317-0200.
Take note that corrosion and fretting can’t be seen outwardly since the parts are implanted in your body. However, there are physical side effects that may point out that your specific hip replacement is malfunctioning. In order to correct the side effects caused by the recalled Stryker implants, you may have to go through a painful revision operation.
You May be Entitled To Compensation
If you received a recalled Stryker hip implant or someone you know did, you need to get a Stryker Orthopedics Hip Replacement Attorney Lawsuit. If you also experienced the painful side effects that may have led to a revision surgery or hospitalization, a Stryker Orthopedics Hip Replacement Attorney Lawsuit is your best chance at getting rightfully compensated. Our Arkansas attorneys are competent at handling malfunctioning medical device cases and will help you to get the compensation you rightfully deserve for your recalled Stryker Orthopedics hip implant.
Contact us today by either phone or e-mail for a free evaluation of your case so we can forge the way forward.