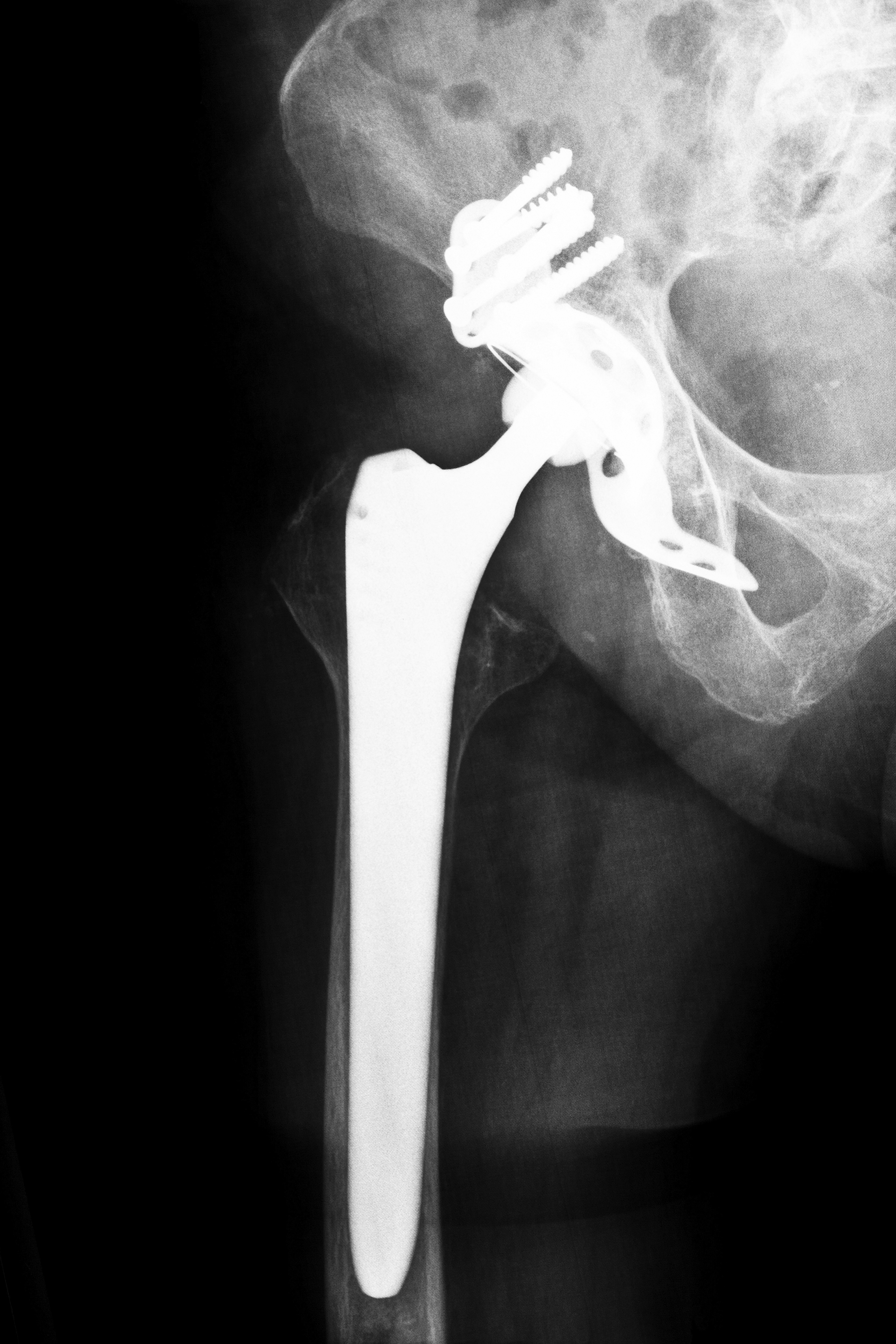
What You Should Know About Biomet M2A Hip Implants
The Biomet M2A Hip replacement device is one of the twenty-one hip implants recalled due to malfunctioning and health risks. The Biomet M2A Hip replacement device is a “metal-on-metal” hip implant, which, like many of comparable devices, was rushed to the market through the FDA 501(K) process without the necessary clinical testing. Since its release in 1998, the FDA has received more than 17,000 cases of malfunction, injury, and harm due to the device and has concluded that the metal-on-metal hip implant poses a greater risk of malfunction than other models. These conclusions came in the aftermath of an unprecedented expert panel in June 2012, which assessed the merits and risks of metal-on-metal hip implants, including the Biomet M2A replacement device. Similarly, medical experts in Canada and Britain produced studies attesting to the unique risks posed by such devices and cautioned against further use of metal-on-metal hip replacements. As such, a number of Georgia Biomet hip lawsuits have ensued.
What Are Side Effects and Risks
If you received a Biomet M2A hip implant during or after 1998, your hip implant is included in the recall of such devices. Many recipients of the Biomet M2A hip replacement device in Georgia report a wide range of complications, some of which are quite serious. Like other metal-on-metal hip replacement devices, the key component in the Biomet M2A hip replacement device is the femoral ball. The femoral ball component is made of chromium and other metals and is prone to grinding excessively against the socket portion of the implant. While plastic femoral ball components may also rub against the socket, the metal femoral ball is prone to irritation and small metal debris may actually chip off from this friction. When this happens, the metal quickly enters the bloodstream and can reach toxic levels. Although extensive clinical trials would have demonstrated this difficulty in the device, the Biomet M2A hip replacement was not subject to clinical trials and was rushed to the market before this complication could be discovered. If you or a loved one has experienced complications after your hip replacement surgery or are currently experiencing complications, you may be eligible for inclusion in current Georgia Biomet hip lawsuits.
 Here is a list of some of the most common adverse side effects and complications associated with the Biomet M2A hip replacement device:
Here is a list of some of the most common adverse side effects and complications associated with the Biomet M2A hip replacement device:
- Constant hip pain
- Tissue death in proximity to joint, or necrosis
- Inflammation or irritation of soft tissue
- Extraordinary muscle pain or muscle sensitivity
- Need for corrective hip surgery or hip revision surgery
- Excessive levels of chromium and other metals in bloodstream
- Symptoms related to metal toxicity (i.e. excessive fatigue, nausea)
- Damage to nerves
- Immobility
- Difficulty walking or increased difficulty moving leg
- Unusual rashes in soft tissue surrounding area
- Unmanageable pain and discomfort
Many individuals experience such symptoms beginning as early as six months after the Biomet M2A hip replacement device was inserted and as late as two years after the procedure. The Biomet M2A hip replacement device recall includes implant procedures performed in 1998 onward.
Pending Legal Action
The Georgia Injury Attorney Group is purusing legal action through Georgia Biomet hip lawsuits. This federal litigation works on behalf of individuals who have experienced hip implant malfunction, loss of mobility, metal toxicity, need for hip revision surgery, pain, suffering, loss of wages, and medical expenses related to Biomet M2A Hip replacement device. Of the over 150,000 hip replacements performed annually, between 5-10% have been the Biomet M2A hip replacement device. Research has shown that this device, like its metal-on-metal replacement device counterparts, is more likely to break down and cause serious, potentially fatal, side effects in patients who have received them. The scope and scale of this law suit is extraordinary as implants included from 1998 to the present are included. In some states, several hundred law suits have been filed and the settlement is expected to include victims throughout the country.
Georgia Biomet hip lawsuits
If you or someone you know has received a Biomet M2A hip replacement device and has experienced complications, you may be eligible to receive compensation for pain, suffering, wages lost due to injury, related medical expenses, and more as a part of these Georgia Biomet hip lawsuits. The Georgia Injury Attorney Group will provide a free and confidential case review, as we are currently filing Georgia Biomet hip lawsuits. The Georgia Injury Attorney Group works with experienced lawyers that bring that experience and sensitivity to each client. Statutes of limitations, which place a limit on how much time an individual has to pursue compensation, may apply, so please contact us as soon as possible.