 What problems are associated with certain metal on metal hip implants?
What problems are associated with certain metal on metal hip implants?
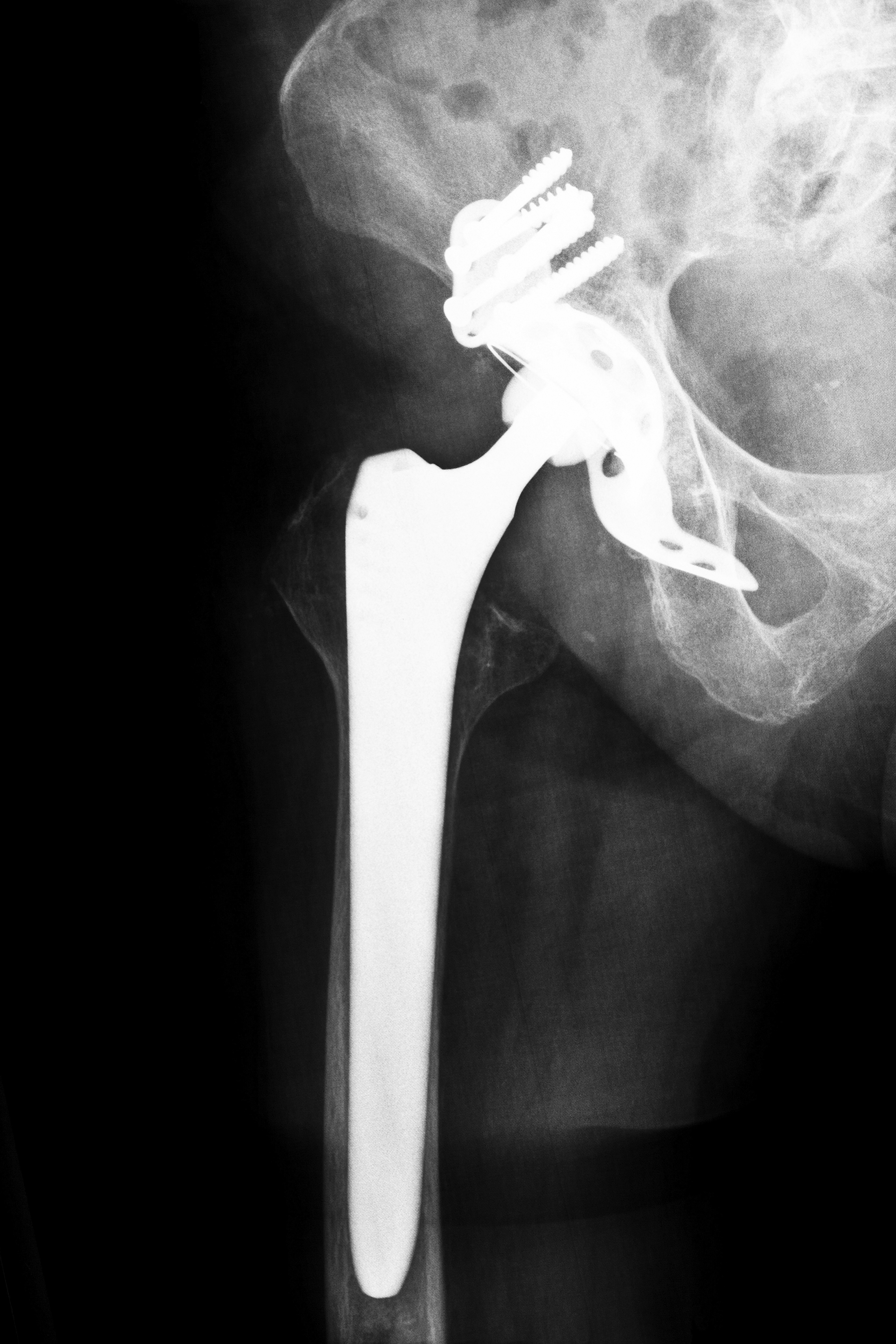
Complaints have often focused on misalignment or friction issues. In a properly functioning artificial hip, the “ball” moves freely and with minimal friction in the “socket.” The ball-and-socket design mimics that of the natural human hip, and it allows excellent freedom of movement when properly deployed.
However, in some metal on metal hip implant systems, undue friction has caused tiny metallic particles to be released into surrounding soft tissue. In some cases, these microscopic particles have made it to a patient’s bloodstream. Manufacturers often used metal alloys in their designs to promote durability. Two potentially toxic elements, chromium and cobalt, were often used in these alloys. Lawsuits now allege that, in certain cases, these elements were released into a patient’s system in sufficient concentrations to cause swelling and/or metal poisoning.
What symptoms have been associated with some metal on metal hip implants?
Some patients have reported serious or severe pain, either in the hip region or in the groin. Fatigue is another reported concern, and it is allegedly related to atomic-size metallic particles making their way into the bloodstream. Other symptoms associated with metal on metal hip implants include limited motion and walking difficulties that allegedly result from a hip joint not moving smoothly. Some patients have reported hip popping or hip squeaking as well.
How can I figure out if my hip replacement system was recalled?
An attorney focused on this area of civil law can assist you in finding this out. It is important to understand that a hip joint does not have to be recalled for a legal claim to be possible. Demonstrable problems may exist in systems that have not yet been recalled. Also, surgical error may be to blame when the ball and socket do not properly align. Undue friction and dislocations may result from misalignment caused by improper technique.
What can be done if my artificial hip is giving me problems?
Surgeons recommend revision surgery in some cases. A problem hip joint is replaced with a second surgery. Although this can alleviate symptoms, a patient still faces another round of recovery. Also, more bone has to be taken in the operation that anchors the ball joint into the bone. This raises the possibility that, years later, subsequent hip surgery might not be possible because of an inadequate length of bone remaining.
Which metal on metal hip implants have thus far been recalled?
A number of manufacturers have been involved in recalls and/or litigation involving their hip systems:
- Smith and Nephew
- Stryker Orthopedics
- DePuy Orthopedics
- Biomet
DePuy Orthopedics is a subsidiary of Johnson & Johnson. Its ASR system is the subject of litigation previously filed in the U.S. District Court in Nashville, Tennessee. About a dozen Tennessee residents joined in the lawsuit filed against DePuy. In part, the lawsuit alleges that DePuy knew of problems with its system in 2007, but it did not issue a recall until 2010.
Stryker has recalled its ABG II and Rejuvenate prosthetic hips that were initially approved by the Food and Drug Administration back in 2008. On July 6, 2012, Stryker voluntarily recalled both medical devices after it received complaints regarding significant pain, hip dislocations and blood poisoning.
Are there deadlines for filing a lawsuit regarding a problem hip implant?
Yes. Like other states, Tennessee state law establishes a set time period from when an injury is first experienced or discovered to when a legal claim must be filed. This is referred to as the statute of limitations.
How can I determine if I have a metal on metal hip implant claim?
Please contact us at Attorney Group for Tennessee. We can answer your questions and connect you with an affiliated attorney who can assist you through the legal process. We provide free consultations to help those who don’t know the next steps they need to take. Contact us today to learn more.