Zimmer Durom Cup Attorneys in Florida
If you have had a Zimmer Durom Cup hip replacement surgery revision or are in need of a revision, you need to speak with Zimmer Durom Cup Attorneys in Florida today. Joint replacement surgery has become increasingly common, with 700,000 such surgeries taking place each year. The increase in joint problems is not just due to an aging population. Because of Americans’ more active lifestyle, people are developing joint problems at a younger age. Strenuous fitness regimens can wear out the major joints, the knees, shoulders and hips, requiring them to be replaced. When younger people need joint replacement, the lifespan of the replacement joints becomes a big issue.
Hip replacement accounts for more than 250,000 joint surgeries every year. Traditionally, replacement hip joints were made of ceramic or plastic, but a recent innovation in hip replacement introduced the use of metal components. They were assumed to have a much longer lifespan than plastic or ceramic artificial hips, so they seemed ideal for younger patients. Metal joints also offered other design benefits, including less wear from rubbing and a decreased chance of breakage or dislocation. However, in the mad rush to get new products to market, some defective products may have been manufactured, sold and implanted into patients.
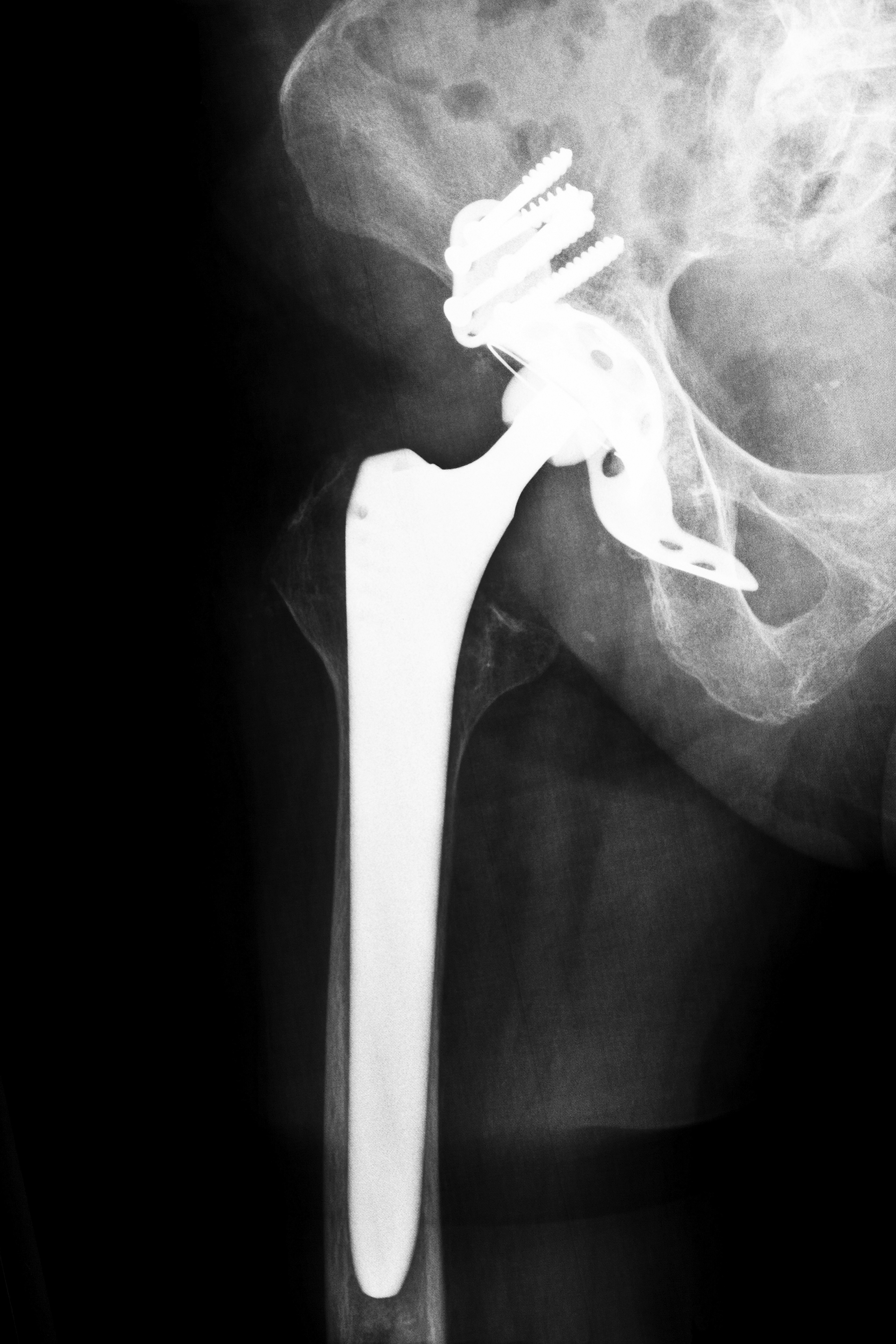
Zimmer Holdings, Inc., a metal hip components manufacturer, made a one-piece metal socket called the Durom Cup. The device had a plasma coating designed to stimulate bones to grow around and onto the implant. The idea was that natural bone would keep the cup in place, rather than the cement that is typically used to hold artificial joints to bones. Between 2006 and 2008, around 12,000 people were implanted with Zimmer Durom Cups. The implants were found to have a high rate of failure, and they were taken off the market and recalled in 2008. Thousands of patients had already suffered problems and needed surgery to have the devices replaced. The recall meant that thousands more would require the additional surgery to have the cups removed and replaced.
Surgeons claimed the reason for the widespread failure of the Zimmer Durom Cup was that the plasma coating didn’t always work properly. Often, the implant would not attach or seal with the bone and would float inside the hip joint. Many patients suffered pain in their hip and back, and some experienced a severe decline in mobility. When surgeons performed repair surgery, they found that some of the implants had no bone growth whatsoever. This could mean the plasma coating itself was potentially defective.
Another possible reason for the failure of the Zimmer Durom Cup was that surgeons were not always properly trained in the correct procedure for implanting the device. Shortly after the recall, the Zimmer Durom Cup was reintroduced to the market with an announcement that the company was working on developing better instructions and training. The implication was that inadequate surgical skill and expertise caused the devices to fail. Of course, a lack of critical surgical training was Zimmer Holdings fault as well. Nevertheless, in June 2010, the New York Times reported that Zimmer was blaming problems with its device on surgical technique rather than any defects in the device itself.
After the 2008 recall, Zimmer Holdings set aside over $270 million for potential lawsuits. The company originally planned to accept claims from replacement surgery that took place within two years of the original surgery. It has revised its position and will now accept claims regardless of how much time elapsed between the two surgical procedures.
Since 2010, several lawsuits have been filed in various jurisdictions against Zimmer Holdings. In early 2013, three more lawsuits were filed by individuals in Idaho, Texas and New York, claiming they had Zimmer Durom Cups implanted and suffered serious injuries as a result. Dozens more law firms around the country, including Florida Zimmer Durom Cup hip recall attorneys, have been examining claims for possible class action and individual product liability suits.
In the state of Florida, as in the rest of the United States, product liability laws protect users of products from harm caused by defects in those products. If you are harmed by a defective product, you can recover damages in the form of reimbursed medical costs, lost wages, compensation for pain and suffering and, potentially, punitive damages. If you are in Florida, and you received a Zimmer Durom Cup and experienced hip pain more than three months after surgery, or your implant loosened or you required replacement surgery, you may be able to recover compensation for your injuries. Florida Zimmer Durom Cup hip recall attorneys can help.
Attorney Group for Florida will help you determine your rights and evaluate your claims. Their dedicated, knowledgeable affiliated attorneys will meet with you to discuss your situation and help you determine whether you can recover damages for your injuries caused by the Zimmer Durom Cup.