Stryker ABG II Modular Neck Stems
The Stryker Corporation markets thousands of products throughout the world and has made in sales over $8 billion in 2012. However, two of their hip implant models have recently been recalled after thousands of patients reported serious complications following implantation. The Stryker ABG II Modular Neck Hip Stem and the Rejuvenate were recalled in July 2012. These recalls are associated with the numerous reported complications linked to the design of metal on metal hip implants that includes the risk of corrosion at the junction of the neck adversely affecting surrounding tissue and bone. Attorney Group for Florida is working with Florida Stryker Hip Recall Lawyers across the state to provide Stryker replacement hip patients with assistance in their hip recall claims.
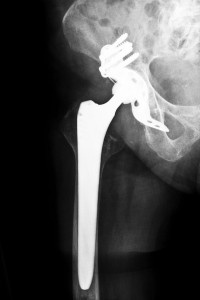
The Stryker ABG II hip replacement system differs from typical hip implant replacement systems in that they include more than just the one piece stem and neck component. Their device is designed with interchangeable necks and stems intended to provide surgeons with more options and more flexibility to custom fit to the needs of each individual patient. The ABG II hip system includes eight left and eight right stems with 10 modular necks designed with the intention to offer better stability and less stress on the bone. Because these parts are made with a mix of iron, zirconium, titanium and molybdenum, the company claims that this particular blend will resist fretting and corrosion due to metal debris accumulating and getting into a patient’s body leading to serious complications. However, data collected after patients began receiving the ABG II modular neck and stem hip system revealed numerous complaints of complications arising from fretting and corrosion after the device was implanted.
Side Effects Associated with the Stryker ABG II Hip System
Even though the numerous amounts of complaints prompted Stryker to issue a recall of its ABG II Modular Hip System, it unfortunately came too late for some recipients of the hip implant. These devices have caused many patients severe side effects, including implant loosening, dislocating and releasing toxic metals into patients’ bloodstreams. When metal toxins spread through a patient’s body, a condition known as metallosis occurs, which can then lead to severe pain, swelling, tissue death, loss of bone, nerve damage, difficulty with cognitive skills, tremors, walking difficulty, damage to other organs and additional surgery—such serious complications that have lead to many lawsuits against Stryker.
Florida Stryker Hip Recall Lawyers
Know your legal options and protect your legal rights. Contact Florida Attorney Group today so that we can match you with experienced Florida Stryker hip recall lawyers to discuss your case. If you or someone you care about had hip replacement surgery and were implanted with one of the recalled Stryker hip replacement systems, you may qualify to seek compensation for your injuries, medical expenses and pain and suffering.





