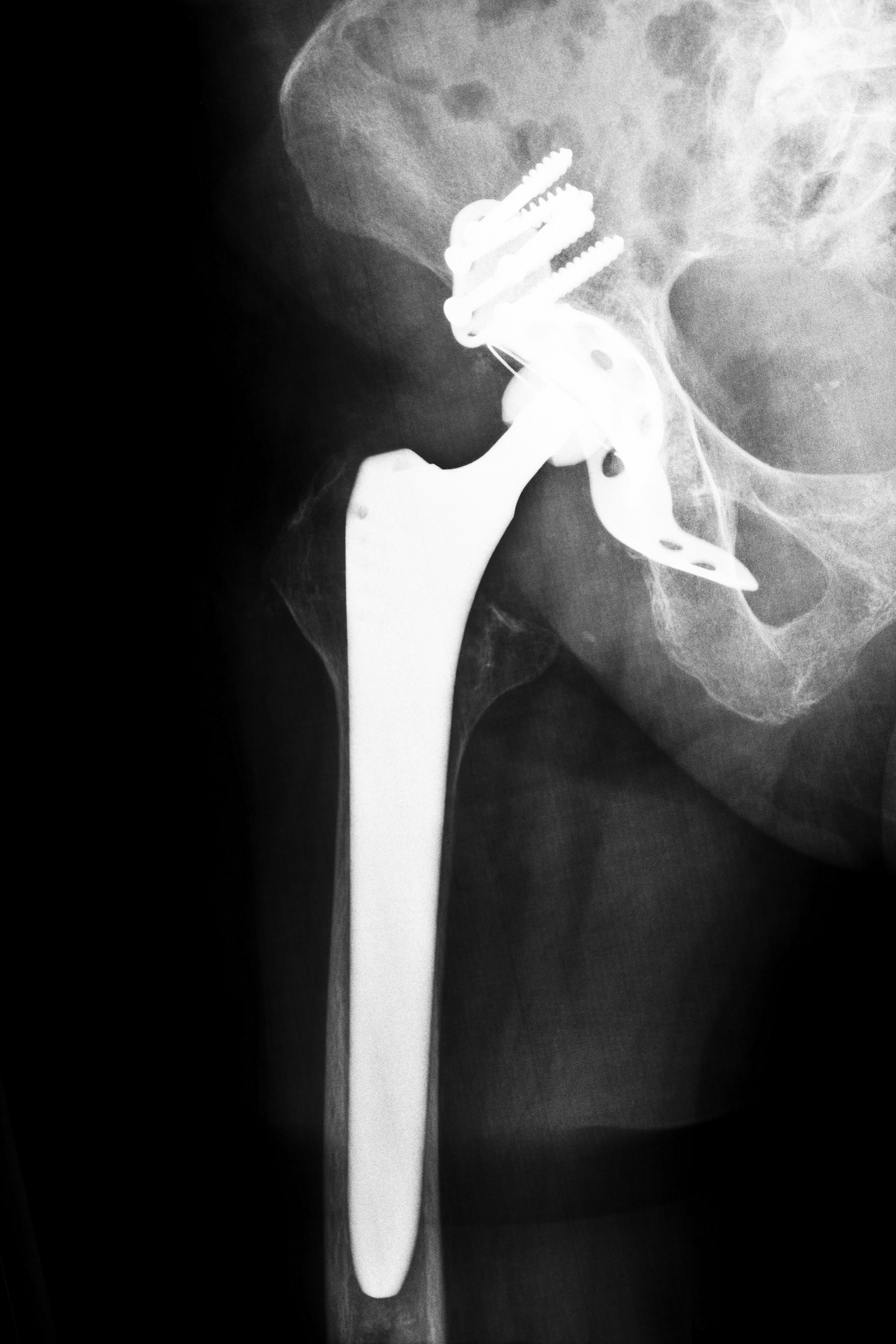
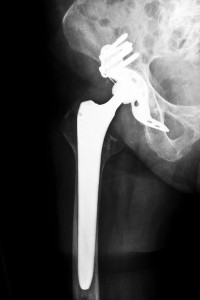
 Manufactured and marketed by New Jersey-based Stryker Orthopaedics, the Rejuvenate modular-neck stem hip implant was recalled on July 6, 2012 due to the potential for the device to corrode at the femoral neck-stem junction. Researchers at the Houston Methodist Hospital in Houston, Texas conducted a study to investigate the rate of early device failure related to corrosion as well as the life expectancy of the Stryker Rejuvenate hip implant. Scientists also intended to analyze the correlation of patient and implant factors with metal ion levels and revision procedures.
Manufactured and marketed by New Jersey-based Stryker Orthopaedics, the Rejuvenate modular-neck stem hip implant was recalled on July 6, 2012 due to the potential for the device to corrode at the femoral neck-stem junction. Researchers at the Houston Methodist Hospital in Houston, Texas conducted a study to investigate the rate of early device failure related to corrosion as well as the life expectancy of the Stryker Rejuvenate hip implant. Scientists also intended to analyze the correlation of patient and implant factors with metal ion levels and revision procedures.
If you or someone you love was fitted with the Stryker Rejuvenate hip implant and subsequently experienced device failure due to corrosion, you may be eligible to work with a hip replacement attorney to seek compensation for your injuries. Contact Attorney Group today to learn more about your legal rights and to consider being connected with an experienced hip replacement attorney.
Corrosion May Lead to Failure of the Stryker Rejuvenate Hip Implant
The study, published on March 19, 2014, took place between June 2009 and July 2012. During this time, 104 patients were fitted with 123 Rejuvenate hip stems by the same surgeon. Following the procedure, researchers measured serum cobalt and chromium levels in each of the patients.
Patients who were experiencing elevated metal ion levels or persistent hip pain underwent magnetic resonance imaging to assess adverse local tissue reactions and osteolysis. Using logistic regression models, researchers were then able to analyze the correlation of implant factors such as femoral head-neck offset, head length and size and stem size as well as patient factors including body mass index, sex and age with the serum metal ion levels they had previously recorded.
After analyzing the data, scientists found that the short-term high rate of corrosion-related revisions after implantation of the Stryker Rejuvenate modular-neck stems was striking. The average duration of follow-up time with the patients was roughly two and a half years. At this point in time, the average cobalt and chromium levels were 5.4 and 2.1, respectively; the differences in metal ion levels between the modular and non-modular groups were significant.
Researchers reportedly determined that 48 percent of the total hip arthroplasties in the group of patients fitted with the modular device led to elevated metal ion levels whereas the metal ion levels in the non-modular group were normal. In participants that were fitted with the modular implant, increased metal ion levels were directly related to a higher femoral head-neck placement as well as a younger age, and both pain and increased cobalt levels indicated a higher risk of requiring revision procedures.
What is the Stryker Rejuvenate Hip Implant?
The Stryker Rejuvenate hip implant may increase a patient’s risk of metal poisoning, pain, swelling, and early device failure. The metal-on-metal parts may potentially corrode and fret, causing shards of debris to accumulate in the patient’s bloodstream and resulting in severe and sometimes life-threatening complications.
In July 2012, Stryker recalled the Rejuvenate and ABG II devices after the company received post-market surveillance data indicating that the metal components could corrode, fret and cause injuries. As a result, patients across the U.S. who were fitted with the Stryker Rejuvenate hip implant are working with a hip replacement attorney to seek compensation for their injuries. A hip replacement attorney can work to ensure those injured receive the maximum compensation to which they may be entitled.
Injured? Consider Seeking Legal Counsel from a Hip Replacement Attorney
Have you or someone you love received the Stryker Rejuvenate hip implant and experienced adverse side effects or complications? Contact Attorney Group today to learn more about working with a hip replacement attorney, pursuing a claim and recovering damages for your condition. We can evaluate your case, free of charge, and can connect you with an experienced hip replacement attorney.