 The first DePuy Pinnacle trial involving the company’s Pinnacle hip implant has come to a close in federal court in Dallas. On October 23, 2014, the jury returned a verdict in favor of the defendants. The trial is first of several scheduled against the medical device maker and its parent company, Johnson & Johnson, in Pinnacle metal hip replacement lawsuits.
The first DePuy Pinnacle trial involving the company’s Pinnacle hip implant has come to a close in federal court in Dallas. On October 23, 2014, the jury returned a verdict in favor of the defendants. The trial is first of several scheduled against the medical device maker and its parent company, Johnson & Johnson, in Pinnacle metal hip replacement lawsuits.
UPDATE: On March 17, 2016, a jury returned a $498 million verdict against the device makers in the second federal bellwether trial.
For more information, contact Attorney Group today. Our consultations are free, confidential and without any obligation on your part.
DePuy Pinnacle Trial Verdict
The hip implant lawsuit, filed on behalf of a patient who was fitted with the device, accused the manufacturer of producing and marketing an allegedly defective product and neglecting to adequately test the implant before offering it to consumers. In this DePuy Pinnacle trial, DePuy marketed the Pinnacle implant and other all-metal hip implants because there was a possibility of high sales, not because they were safer or more effective than other metal-on-metal products currently on the market.
The DePuy Pinnacle trial is the first of many, and the end result of this case does not necessarily predict the outcome of future cases.
UPDATE:
On March 17, 2016, a Dallas Jury in the second DePuy Pinnacle metal hip bellwether trial handed down a $498 million verdict against Johnson & Johnson and its DePuy subsidiary. The award included $360 million in punitive damages against the device makers.
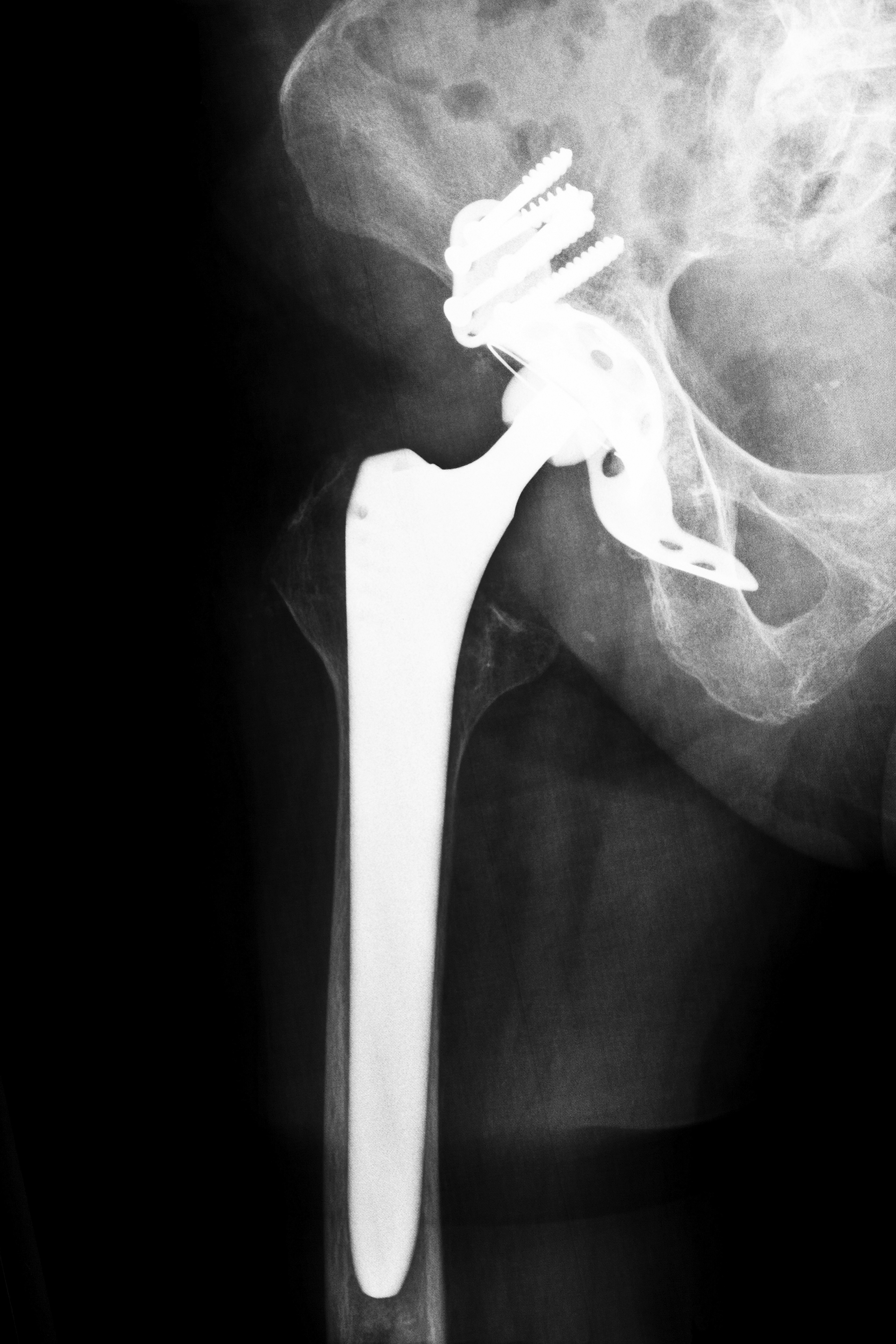
What is the Pinnacle Hip Implant?
The DePuy Pinnacle was designed to provide a greater range of motion and reduced pain for patients living with hip complications. However, the device’s all-metal components allegedly caused recipients to suffer from several adverse complications, including difficult walking, dislocation, clicking noises from the joint, decreased range of motion, and increased pain.
Additionally, the implant allegedly causes metal ions to accumulate in patients’ bloodstreams, causing a condition known as metallosis, or metal poisoning. As a result, many Pinnacle recipients suffered from early device failure and required painful and costly revision procedures to remove the allegedly defective device.
More than 6,500 Pinnacle lawsuits have been filed in a multidistrict litigation that is currently underway in Dallas federal court. Patients accuse DePuy and its parent company, Johnson & Johnson, of defectively designing the product, gross negligence, and a failure to warn patients of the potential complications.
Those who believe that they have a Pinnacle lawsuit are urged to contact DePuy Pinnacle Hip Replacement Attorney to learn more about their rights and remedies.
Contact Us For More Information
For more information, contact Attorney Group. You can fill out the form on this page, call us at the number listed at the top of the page, or email us at info@attorneygroup.com.
When you contact us, an attorney will follow up with you to speak with you about your case or answer questions that you might have. There is no cost or obligation to speak with us, and any information you provide will be kept confidential.
Please note that the law limits the time you have to pursue a claim or file a lawsuit for an injury. If you think you have a case, you should not delay taking action.
See our Frequently Asked Questions page for more information, and contact Attorney Group today.